From Urine to Neurons & Ancient Herbs to Nano-Delivery

On December 19th, 2025, I had the privilege of attending the International Conference on Integrative Biosciences and Translational Research (ICIBTR 2025). Hosted in a hybrid mode at the Noorul Islam Centre for Higher Education (NICHE) in Kanyakumari, Tamil Nadu, the event was organized by the Department of Allied Health Sciences in association with the NIMS Centre for Genomic Medicine, Thiruvananthapuram.
The core focus of the conference was the "bench-to-bedside" continuum—bridging the gap between basic bioscience research and real-world clinical applications. The sessions were incredibly insightful, focusing heavily on cutting-edge genetics, stem cell therapy, and neurodegenerative disease treatments.
Here is a detailed breakdown of the key sessions and my personal takeaways from the event.
Session 1: The Future of Regenerative Medicine – Stem Cells and Reprogramming
Speaker: Prof. Dr. Johnson Rajasingh (Professor, The University of Tennessee Health Science Center, USA)
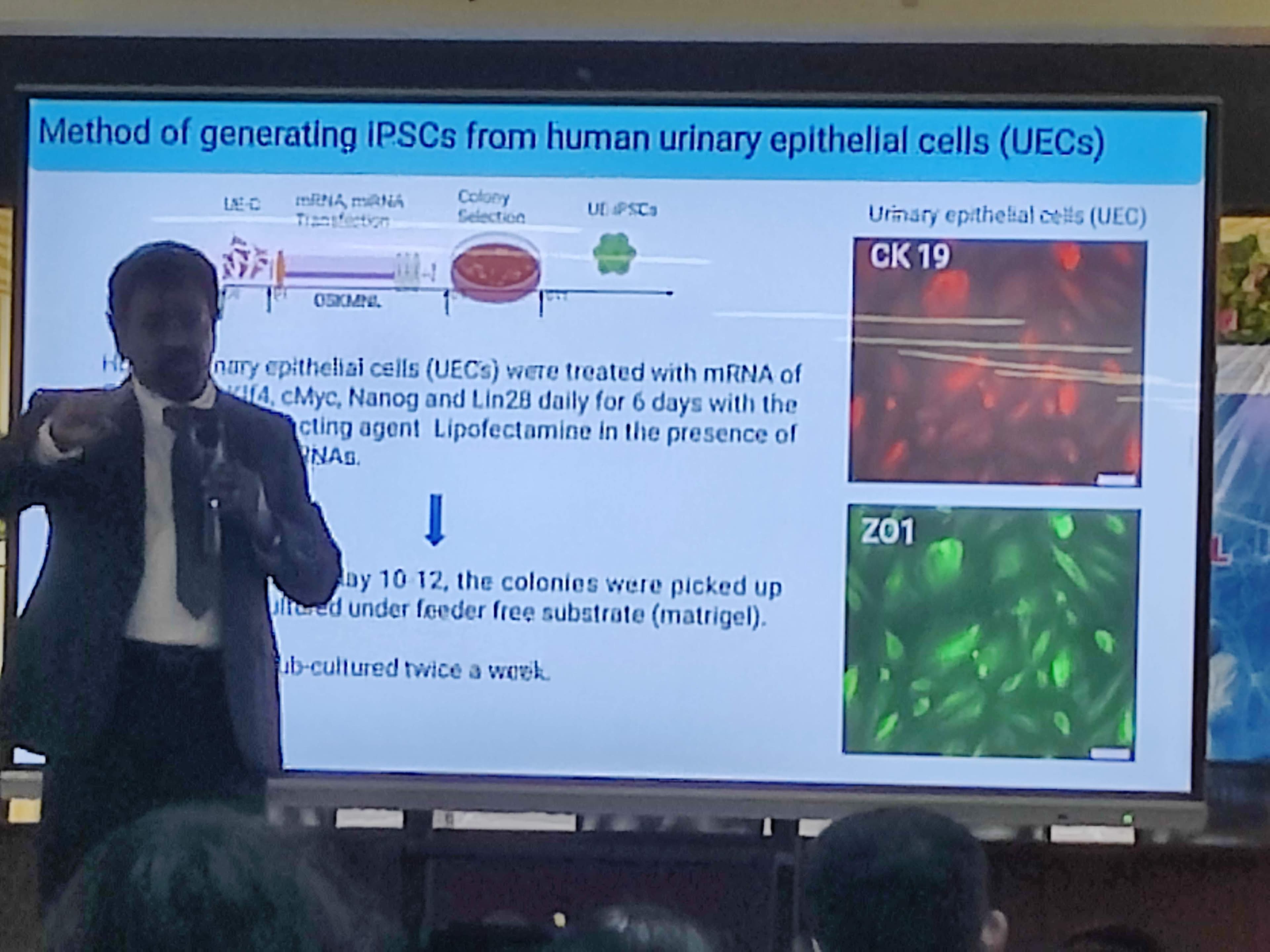
Dr. Rajasingh’s session dove deep into the fascinating world of cellular regeneration, specifically focusing on Induced Pluripotent Stem Cells (iPSCs) as an ethical and highly effective alternative to Embryonic Stem Cells (which face strict legal and ethical bans in places like the USA).
To set the stage, he provided a clear hierarchy of stem cells:
- Totipotent Cells: Cells with the ultimate potential to produce an entire living organism.
- Pluripotent Cells: Extracted from the inner cell mass of a day-5 embryo. They can differentiate into almost any cell in the body, but harvesting them destroys the embryo.
- Multipotent Cells: Cells with limited capabilities (e.g., they can differentiate into blood cells, but not heart or brain cells).
- Unipotent Cells: Resident cells committed to a single lineage, like specific skin cells.
The Magic of iPSCs: Reprogramming Somatic Cells
Since every cell in our body contains the same embryonic genes (though only specific ones are active depending on the cell type), the goal of regenerative medicine is to "wake up" the dormant genes in a normal adult somatic cell.
Historically, this was attempted via oocyte-dependent nuclear reprogramming (replacing the nucleus of an egg cell to create a clone), but this is banned in humans. Instead, researchers developed a method using viruses to reprogram cells.
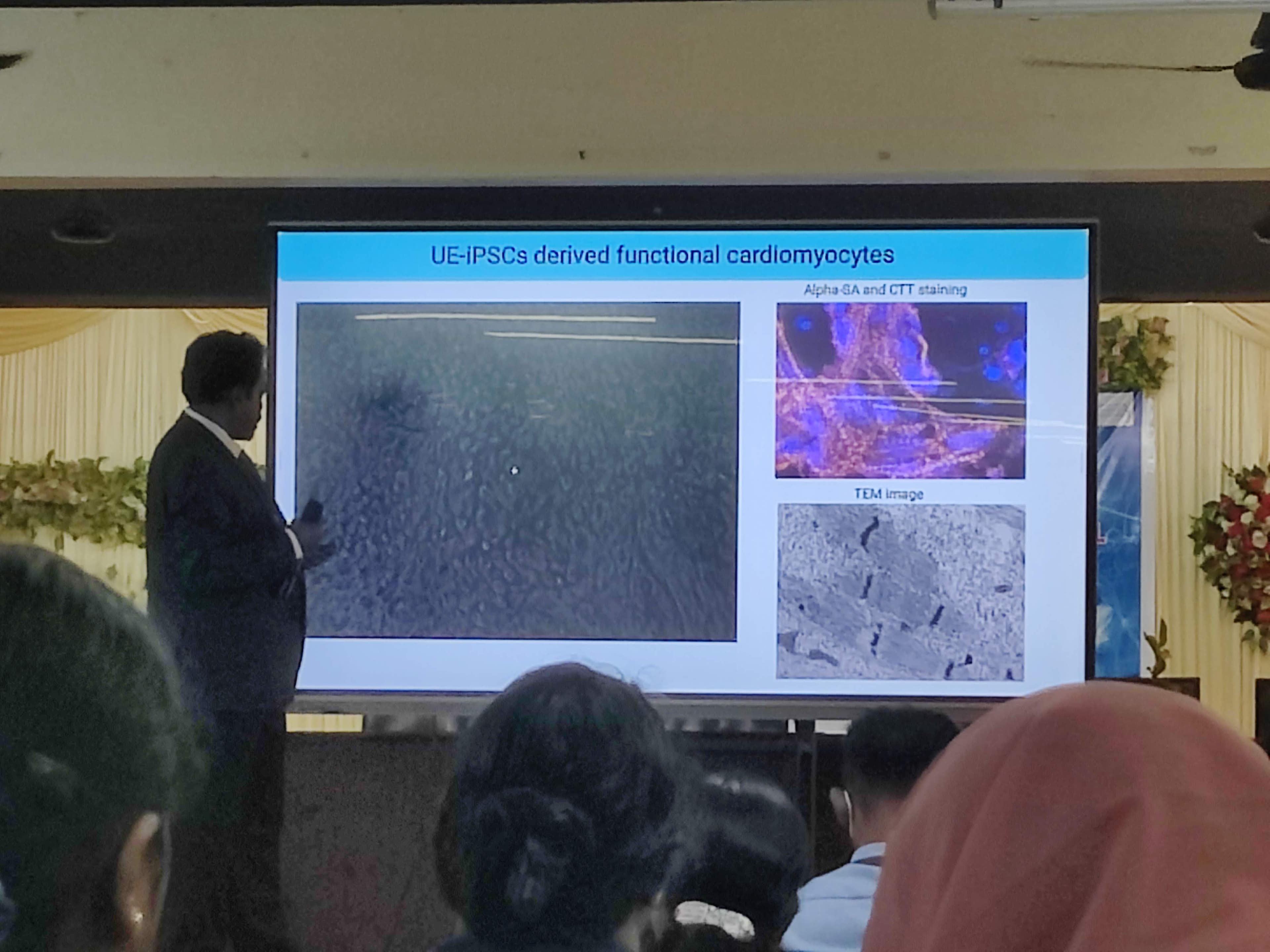
Dr. Rajasingh detailed a groundbreaking method where Human Urinary Epithelial Cells (UECs) are treated daily for 6 days with an mRNA cocktail containing specific factors: Oct4, Sox2, Klf4, Nanog, and Lin28. This successfully activates the inactive embryonic genes, generating iPSCs! He showcased how these cells can then be directed to differentiate into blood cells, hepatocytes (liver cells), and even nerve cells.
Clinical Applications & Exosomes:
- Better than Umbilical Cord Storage: We may no longer need to pay for expensive, decades-long storage of umbilical cord mesenchymal stem cells. iPSCs can be differentiated into mesenchymal stem cells on demand and actually show better wound-healing properties.
- Sports Medicine: These cells show massive promise for treating sports injuries by generating fresh osteocytes and chondrocytes.
- Exosomes: Dr. Rajasingh also touched on exosomes—tiny vesicles (around 150nm) used for cell-to-cell communication. Researchers are currently studying how to "tune" somatic cells to secrete disease-specific exosomes to treat conditions like Parkinson's and heart attacks.
The Catch: The technology is still evolving. Currently, the reprogramming process has a high mortality rate for the cells (about 95% die, leaving a 5% survival rate). Furthermore, while the engineered cells look and act almost identical to natural cells, they cannot perfectly replace every single function of a real cell yet.
Session 2: Revolutionizing Alzheimer's Disease Care
Speaker: Dr. Ashok Iyaswamy (Research Assistant Professor, School of Chinese Medicine, Hong Kong Baptist University)
Dr. Iyaswamy delivered a captivating presentation on the diagnostic and therapeutic potential of engineered exosomes for Alzheimer's Disease (AD). He started by highlighting a grim reality: most AD drugs that work in primate models fail in humans. Furthermore, current targets like Tau proteins and Amyloid-beta (Aβ) plaques are merely factors of the disease; current medicines only offer symptomatic relief, while a true cure remains elusive.
From Traditional Chinese Medicine (TCM) to Molecular Science
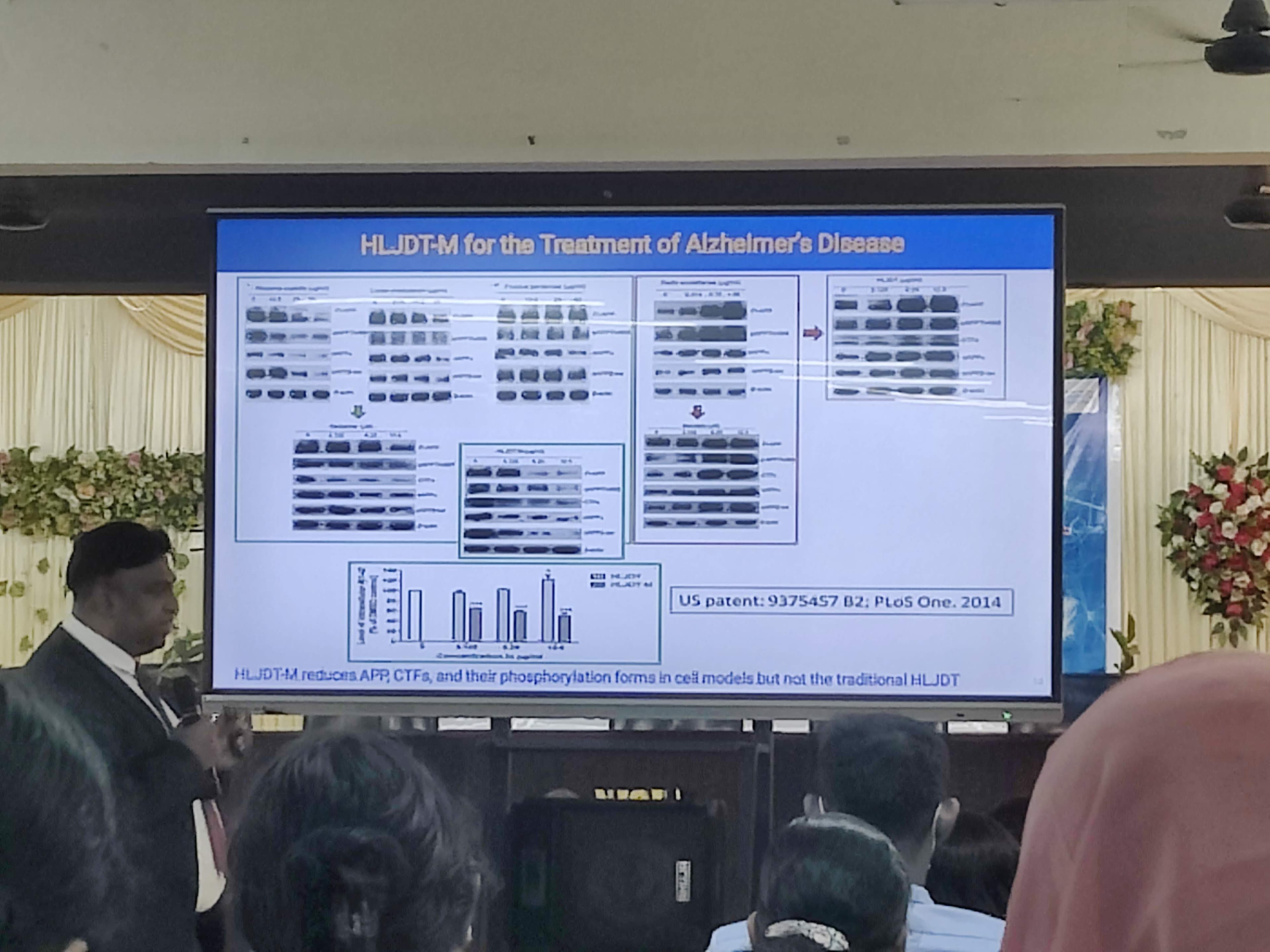
Dr. Iyaswamy’s team looked to ancient literature for answers. They investigated a traditional Chinese formulation called Huanglian-Jie-Du-Tang (HLJDT). During their research, they found that one specific constituent actually mimicked Alzheimer's symptoms. By isolating and removing that specific compound, they created a highly effective modified version called HLJDT-M, which has now been patented.
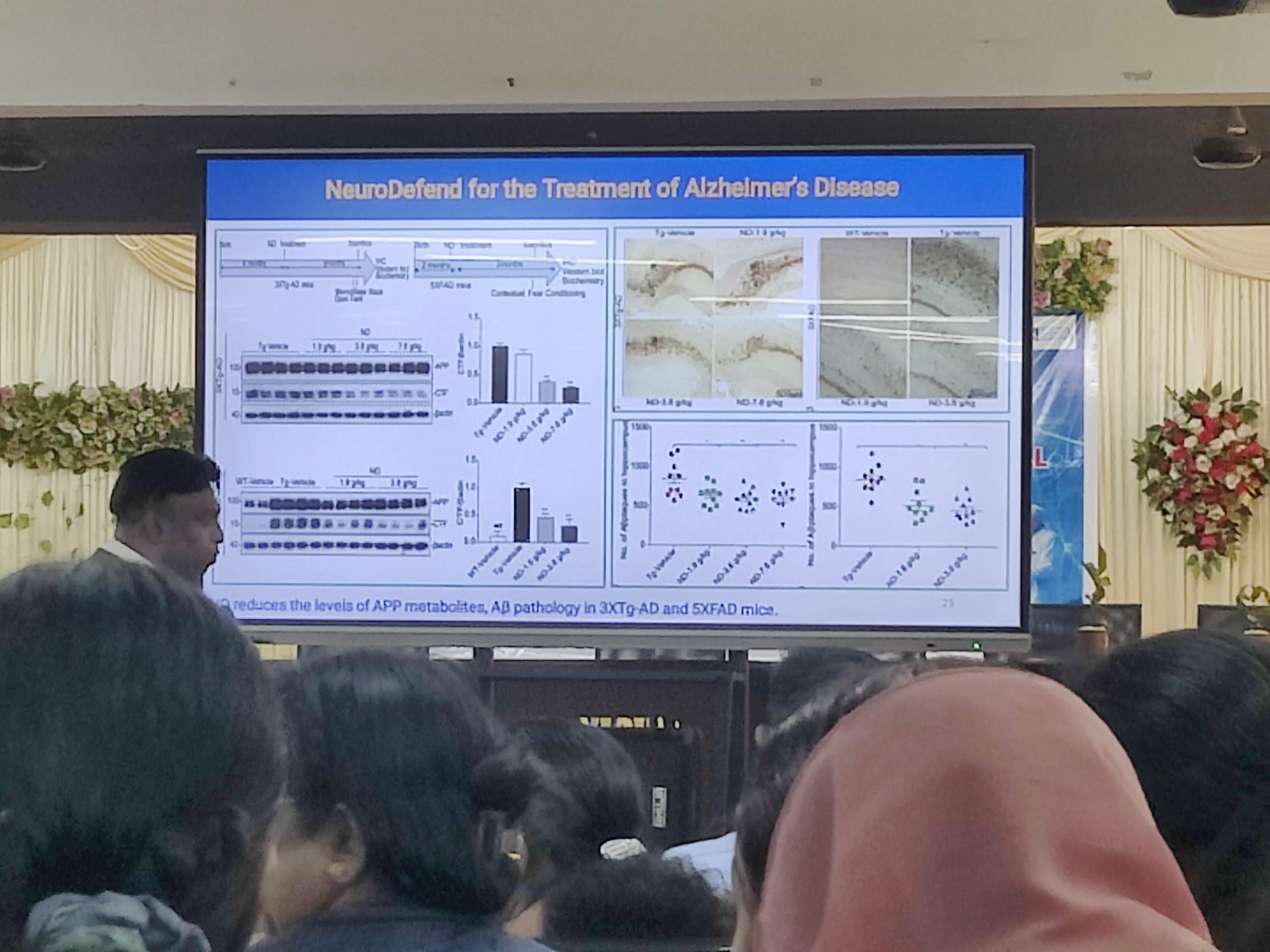
Building on this, they developed a new formulation called NeuroDefend, containing six specific herbs. In animal models, it was shown to significantly reduce insoluble Tau proteins and improve cognitive function.
Triggering Autophagy and Crossing the Blood-Brain Barrier
A major issue in Alzheimer's is autophagy dysfunction—where the brain's waste-clearing mechanism (via lysosomes) fails to clear out toxic proteins.
Dr. Iyaswamy introduced a compound called F-SLOH. Initially used as a diagnostic tool because it binds to Aβ plaques and fluoresces under imaging, the researchers realized it also had therapeutic potential. F-SLOH successfully triggers autophagy by activating the TFEB transcription factor (via MAPK1 inhibition) and increasing PP2A expression. It was shown to actively recruit microglia to help clear the plaques.
However, treating the brain requires crossing the highly restrictive Blood-Brain Barrier (BBB). To solve this, Dr. Iyaswamy's team engineered Fe65-Exosomes derived from hippocampal neurons. Because Alzheimer's patients already express the Fe65 protein, these engineered exosomes act as a perfect "Trojan horse" vehicle to deliver therapeutics directly into the brain via IV or IP injection.
The Results: He claims to be the first in the world to achieve this specific exosomal delivery. Mouse models showed high brain uptake, low toxicity, improved motor and cognitive abilities, and enhanced new synapse formation.
My Key Takeaway & Q&A Experience
At the end of Dr. Iyaswamy's session, I had the opportunity to ask him a question that had been on my mind: "What are the main difficulties faced when taking Traditional Medicine and translating it into modern Molecular Medicine?"
His answer was immediate and grounded in reality: Funding. He explained that taking ancient, multi-compound herbal remedies and putting them through the rigorous, highly controlled pipelines of modern molecular isolation, animal modeling, and clinical trials requires an immense amount of capital. It was a great reminder that behind every scientific breakthrough is an ecosystem of grants, investors, and immense financial risk.
Conclusion
Attending ICIBTR 2025 was a phenomenal experience. From the ethical workarounds in stem cell generation to the marriage of ancient herbal medicine and cutting-edge nanotechnology, the "bench-to-bedside" approach is clearly the future of global healthcare. The research presented here offers real hope for conditions that were previously thought to be untreatable.
Gallery

NICHE Campus Entrance

Main Conference Hall

Exploring NICHE campus
Scientific Session in progress
Scientific Session

Scientific Session

Scientific Session
Scientific Session
Scientific Session

Scientific Session

Scientific Session